Alpha-Lipoic Acid: The Science Behind the 65% Search Surge
**Meta Description:** Learn the truth about alpha-lipoic acid: search +65%. Discover what the surge in searches reveals about this powerful antioxidant and how to find quality supplements. --- You're scrolling through health forums at 2 AM, coff...

D.C.
Doctor of Chiropractic
Founder of FormulaForge. Doctor of Chiropractic revolutionizing nutritional supplements to be what we all wish they were — real quality, scientifically validated, with the exact ingredients and dosages we choose.
View Full ProfileReviewed by Dr. Brennan Commerford, DC
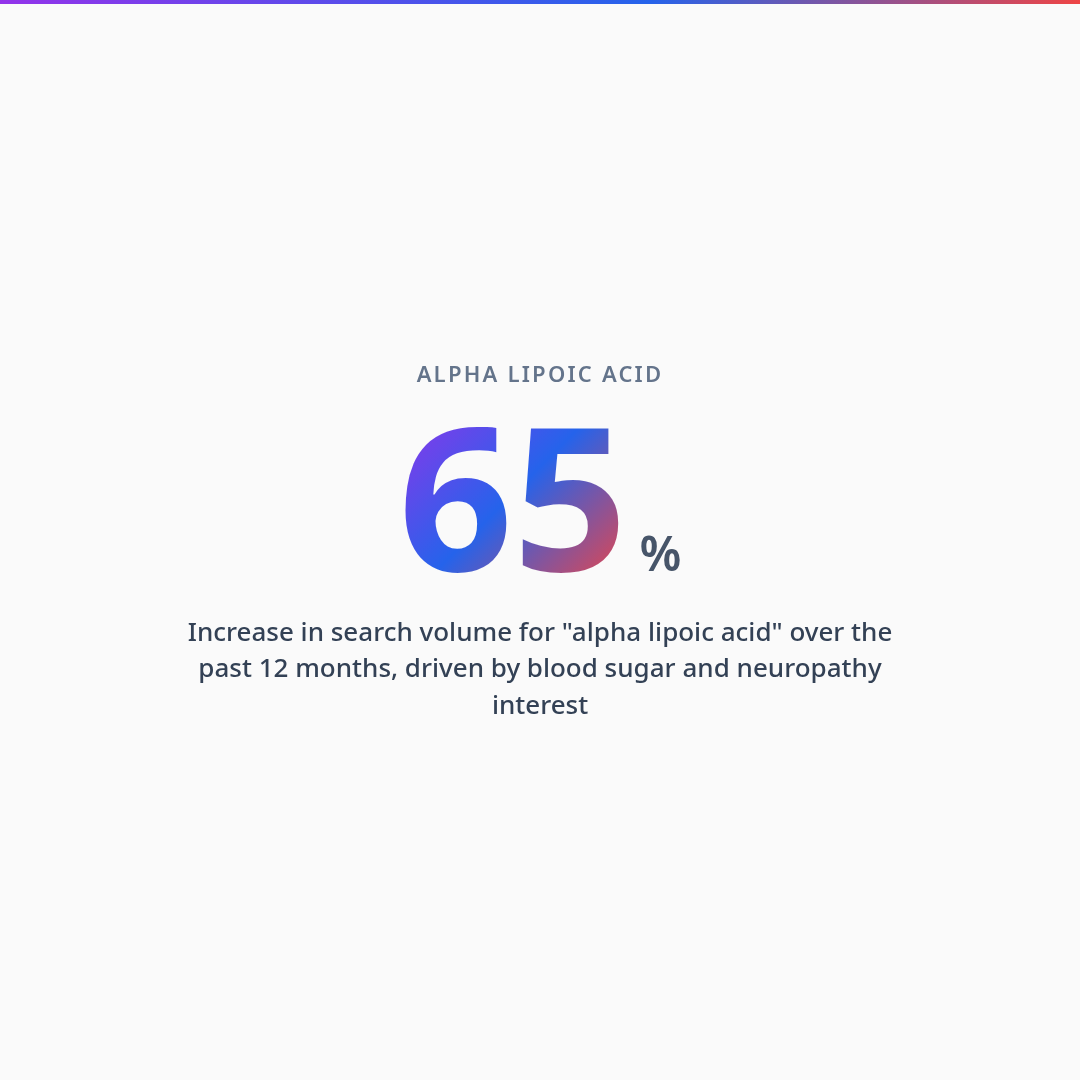
Alpha-lipoic acid has seen a 65% search volume surge in the past year — driven by interest in its antioxidant recycling properties, blood sugar support, and neuroprotective effects. But most ALA supplements sold in the US use the racemic (R+S) form, which delivers only 50% of the biologically active R-isomer. This guide covers why the R vs. S distinction matters, how ALA functions as the "master recycler" of the antioxidant network, the clinical evidence for blood sugar and nerve health applications, and how to read a label to ensure you are getting a supplement that actually works.
What Is Alpha-Lipoic Acid?
Alpha-lipoic acid (ALA) is a sulfur-containing compound that functions as an essential co-factor in mitochondrial energy metabolism — specifically in the pyruvate dehydrogenase and alpha-ketoglutarate dehydrogenase complexes, two critical enzymes in the Krebs cycle. Unlike most micronutrients, ALA is both water-soluble and fat-soluble, meaning it can operate in both aqueous cellular compartments and lipid membranes. This dual solubility is one of the properties that makes it uniquely effective as an antioxidant.
The body produces small amounts of ALA endogenously, but production declines with age. Food sources (spinach, broccoli, organ meats) contain meaningful amounts but are protein-bound and have significantly lower bioavailability than supplemental free ALA. The clinical applications that have attracted the most interest — neuropathy, insulin sensitivity, antioxidant network support — require doses far above what dietary sources can deliver.
Alpha-lipoic acid was originally classified as a vitamin in the 1950s before it was discovered that the body could synthesize small amounts. It was then reclassified as a vitamin-like compound. The endogenous synthesis pathway requires several B vitamins and trace minerals, which means deficiencies in these co-factors can impair ALA production — one of the reasons B-complex status is relevant context when assessing need for ALA supplementation.
The Antioxidant Recycling Network: Why ALA Is Called the "Master Recycler"

Most antioxidants are consumed when they neutralize a free radical — they donate an electron, become oxidized themselves, and require external replenishment. ALA occupies a unique position in this network because it can regenerate other antioxidants after they are oxidized, effectively extending their useful lifespan. This is what earns ALA its "master antioxidant" or "master recycler" designation.
Vitamin C (ascorbate) donates electrons to neutralize free radicals and becomes dehydroascorbate — an oxidized, inactive form. Vitamin E (alpha-tocopherol) neutralizes lipid peroxidation chain reactions and becomes a tocopheryl radical. Glutathione, the primary intracellular antioxidant, becomes GSSG (oxidized glutathione). Without regeneration, these antioxidants are consumed faster than they are replenished under oxidative stress.
ALA and its reduced form, dihydrolipoic acid (DHLA), can directly regenerate Vitamin C from dehydroascorbate, regenerate Vitamin E from the tocopheryl radical, regenerate glutathione by reducing GSSG, and chelate transition metals (iron, copper) that catalyze free radical chain reactions. This network effect means ALA supplementation amplifies the antioxidant activity of multiple other compounds simultaneously — including those already present in your diet.
Packer et al. published a landmark review in Free Radical Biology and Medicine (1995) establishing the "antioxidant network" concept and ALA's central role in it. Subsequent research confirmed that DHLA (the reduced form of ALA) has broader antioxidant reach than ALA itself — it can directly neutralize reactive oxygen species in both aqueous and lipid environments, regenerate Vitamin C and E, and maintain intracellular glutathione levels. A 2012 review in Antioxidants & Redox Signaling quantified that DHLA effectively amplifies the antioxidant capacity of the network by a factor of 3–5x compared to ALA alone.
R-ALA vs. S-ALA: The Form That Actually Matters
This is the most important clinical distinction in ALA supplementation and the one most often omitted from product labels.
ALA exists as two mirror-image molecular structures — the R-isomer (R-ALA) and the S-isomer (S-ALA). In nature, only the R-form exists — it is what the body produces endogenously and the form that functions as a co-factor in mitochondrial enzymes. The R-form is the biologically active isomer.
The S-form is an artifact of chemical synthesis. Most ALA supplements are produced through a racemic synthesis process that yields a 50/50 mixture of R and S forms — labeled as "alpha-lipoic acid" or "racemic ALA." This means that the majority of ALA products on the market deliver only 50% of the active compound per stated dose. A 600 mg racemic ALA supplement delivers 300 mg of R-ALA.
If a label says "alpha-lipoic acid" without specifying "R-ALA," it is almost certainly the racemic 50/50 mixture. Na-R-ALA (sodium R-lipoate) is the most bioavailable form and requires lower doses for equivalent effect. Racemic ALA is not ineffective — the research on blood sugar and neuropathy was conducted largely on racemic ALA — but R-ALA and Na-R-ALA represent meaningful upgrades in delivery.
Blood Sugar Support: The Clinical Evidence
ALA's application in blood sugar regulation is one of its best-studied areas, particularly in the context of type 2 diabetes and metabolic syndrome. The mechanisms are multiple: ALA activates GLUT4 translocation to the cell surface (improving glucose uptake independently of insulin), reduces oxidative stress that impairs insulin receptor signaling, and improves mitochondrial function in insulin-sensitive tissues.
The ALADIN III (Alpha-Lipoic Acid in Diabetic Neuropathy) trials are among the most important ALA studies. ALADIN III tested intravenous ALA in diabetic neuropathy patients and demonstrated significant improvements in neuropathy symptoms, but the oral trials also generated meaningful data. A 2011 meta-analysis in Annals of the New York Academy of Sciences reviewed 9 RCTs of oral and IV ALA in type 2 diabetes patients and found research has explored the relationship between ALA supplementation and markers of healthy blood sugar metabolism. Effect sizes were modest but consistent across studies.
Diabetic Neuropathy: The Strongest Clinical Application
ALA has been extensively studied in clinical settings for nerve health applications — chronic nerve damage from elevated blood glucose that causes pain, numbness, and tingling, primarily in the feet and legs. The ALADIN, SYDNEY, and NATHAN trials collectively form one of the most robust evidence bases for any nutritional intervention in this condition.
The SYDNEY 2 trial (2006) was the pivotal oral ALA neuropathy study: A clinical trial examined 600 mg/day ALA over 5 weeks and reported improvements in participant-reported nerve comfort outcomes compared to placebo (p less than 0.001). A 2012 meta-analysis in European Journal of Endocrinology of 15 trials involving 1,258 patients found that both IV and oral ALA significantly reduced neuropathy symptoms. The effect size for intravenous ALA was larger, but oral 600 mg/day showed clinically meaningful improvements. ALA has been studied extensively in European clinical settings for nerve health, where it has been used clinically since the 1960s.
Other Applications with Evidence Support
A 2017 meta-analysis in Obesity Reviews (12 RCTs, 545 participants) found that Some research has examined the relationship between ALA supplementation and body composition markers and BMI versus placebo. The effect is modest but real. Proposed mechanisms include AMPK activation and reduced appetite signaling. Doses in trials ranged from 300–1800 mg/day; larger effects were seen at higher doses.
ALA crosses the blood-brain barrier and reduces oxidative stress in neural tissue. Preliminary research has explored ALA in the context of supporting healthy cognitive function in older adults over 12–48 months in one observational study. Controlled RCT data specifically for cognitive outcomes is limited. The mechanism is plausible given ALA's ability to regenerate neural antioxidants and improve mitochondrial function in neurons, but clinical confirmation in humans is incomplete.
In vitro data and some animal studies suggest high-dose ALA may reduce T3 levels and compete with thyroid hormone transport. The clinical significance in humans at standard doses (600 mg/day) has not been confirmed, but individuals with hypothyroidism or on thyroid medication should monitor thyroid function with ALA supplementation and discuss with their prescribing physician. This is a meaningful precaution for a population that frequently uses ALA.
Dosing: Standard vs. R-Specific
Individuals with diagnosed conditions should consult their physician.
The FormulaForge formulary distinguishes between racemic alpha-lipoic acid and R-ALA (including Na-R-ALA) as separate tier entries with different dose equivalence calculations. When you analyze a supplement containing ALA at myformulaforge.com, the system identifies which isomeric form is present, adjusts the effective R-ALA dose accordingly, and cross-references against clinical trial doses for your stated health goal. If you are taking 600 mg racemic ALA for neuropathy, the system confirms this aligns with the SYDNEY 2 trial protocol. If you are paying a premium for R-ALA, the system confirms you are within the equivalent active dose range.
ALA can lower blood glucose — this is a feature of its mechanism, not just a side effect. Patients taking insulin, sulfonylureas, or other glucose-lowering medications must monitor blood glucose carefully and coordinate ALA supplementation with their prescribing physician to avoid hypoglycemia. Na-R-ALA absorbs very rapidly and can produce a transient glucose drop — it should always be taken before a meal, not on a fully empty stomach. ALA may reduce the effectiveness of some chemotherapy agents by reducing oxidative stress in cancer cells; patients undergoing chemotherapy should discuss with their oncologist. At doses above 600 mg/day, nausea and GI discomfort are the most common adverse effects.
Frequently Asked Questions
Alpha-lipoic acid has its strongest clinical evidence for diabetic peripheral neuropathy — where the ALADIN and SYDNEY trial series represent some of the most robust nutritional intervention data for any nerve condition. Blood sugar and insulin sensitivity support has moderate RCT evidence. The antioxidant recycling mechanism is genuine and meaningful. The key purchasing decision is form: racemic ALA is used in most research and is effective, but R-ALA (particularly Na-R-ALA) delivers the same active compound at lower doses with better bioavailability. If the label says "alpha-lipoic acid" without the R prefix, you are getting 50% active isomer. For neuropathy and blood sugar applications at therapeutic doses, this distinction is clinically relevant.
This article is intended for educational and informational purposes only and does not constitute medical advice. The information provided here is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the guidance of your physician or other qualified health provider with any questions you may have regarding a medical condition or supplement regimen. These statements have not been evaluated by the Food and Drug Administration. Dietary supplements are not intended to diagnose, treat, cure, or prevent any disease.
- Ziegler D, et al. Treatment of symptomatic diabetic peripheral neuropathy with the antioxidant alpha-lipoic acid: a 3-week multicentre randomized controlled trial (ALADIN Trial). Diabetologia. 1995;38(12):1425–1433.
- Ziegler D, et al. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care. 2006;29(11):2365–2370.
- Packer L, et al. Alpha-lipoic acid as a biological antioxidant. Free Radic Biol Med. 1995;19(2):227–250.
- Gu L, et al. Effect of alpha-lipoic acid on lipid metabolism in individuals with obesity. Eur J Pharmacol. 2017;798:1–10.
- Koh EH, et al. Effects of alpha-lipoic acid on body weight in obese subjects. Am J Med. 2011;124(1):85.e1–8.
- Hager K, et al. Alpha-lipoic acid as a new treatment option for Alzheimer's disease — a 48 months follow-up analysis. J Neural Transm. 2007;72(Suppl):189–193.
- Shay KP, et al. Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta. 2009;1790(10):1149–1160.



Related Research
- ResearchGalactooligosaccharides (GOS) (Prebiotic) — Forms, Dosing & Research(1 form)
- ResearchL-Citrulline DL-Malate (different ratio) — Forms, Dosing & Research(1 form)
- ResearchALA (Alpha-Linolenic Acid) Flax Oil — Forms, Dosing & Research(1 form)
- ResearchMagnesium (as DiMagnesium Malate) — Forms, Dosing & Research(1 form)

